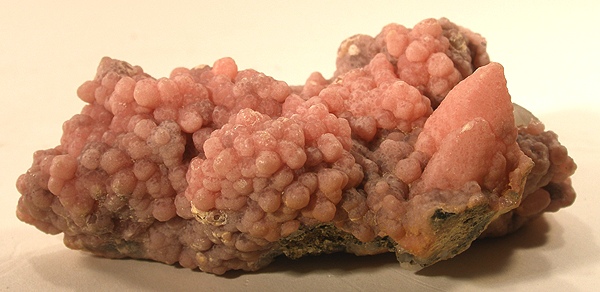
Crocoite
Crocoite
PbCrO₄ Properties
- Category
- Mineral
Crocoite is a lead chromate that forms splendid prismatic crystals of intense red-orange color, so rare and sought after by collectors that they are considered among the most beautiful minerals in the world. It forms in oxidizing environments of lead deposits and represents an advanced stage of supergene alteration.
Crocoite (PbCrO₄) is a secondary mineral that originates exclusively in supergene alteration profiles of lead deposits, where oxygen-rich solutions and meteoric waters interact with primary minerals containing lead and chromium. Its formation requires very specific geochemical conditions: oxidizing environments, moderately acidic pH, and the simultaneous availability of chromate ions, which is not frequent in nature. Crystals, which can reach several centimeters in length, display characteristic adamantine luster and transparency ranging from translucent to transparent. From a crystallographic perspective it belongs to the monoclinic system with point group 2/m, and crystals commonly exhibit elongated prismatic forms along the c-axis.
The most significant mineralogical associations include cerussite (PbCO₃), anglesite (PbSO₄), smithsonite (ZnCO₃), and other oxides and carbonates from oxidation zones. Crocoite was first identified in Tasmania in 1832 and remains synonymous with gemological and collecting excellence precisely because of Tasmanian crystals, which still represent the worldwide standard for quality and size. Gem-quality specimens are extraordinarily rare and command elevated prices in the international collecting market.
Crocoite: PbCrO₄, space group P2₁/n (monoclinic), lattice parameters a = 7.14 Å, b = 7.41 Å, c = 6.81 Å, β = 102.5°. Hardness 2.5–3 Mohs, density 5.97–6.10 g/cm³. Refractive indices nα = 1.820, nβ = 1.835, nγ = 1.920; birefringence Δ = 0.100 (strong), optical sign negative. Red-orange color due to electronic transitions of the chromate ion (CrO₄²⁻), which exhibits a characteristic absorption band around 540 nm. Imperfect cleavage along plane (010), conchoidal fracture. Adamantine luster. Fluorescence absent or very weak under UV. Soluble in dilute acids with liberation of chromic acid. Raman spectroscopy shows characteristic bands of chromate vibrational modes around 850 cm⁻¹ (symmetric stretching) and 900 cm⁻¹ (asymmetric stretching). Crystals commonly prismatic along the c-axis, with secondary forms (110), (100), (010). Mineralogical associations: cerussite, anglesite, malachite, limonite, goethite. Alteration: crystals may undergo dehydration and transformation into amorphous phases if exposed to moisture and direct light for prolonged periods.
Mining localities
- Dundas, Tasmania, Australia
- Sverdlovsk, Urali, Russia
- Beresov, Urali, Russia
- Mammoth Mine, Arizona, USA
- Tsumeb, Namibia
- Wölsendorf, Baviera, Germania
- Callao, Perù
Frequently Asked Questions
Where is crocoite found and in which locations is it most common?
Crocoite is found mainly in oxidation environments of lead deposits, with the finest specimens coming from Australia (Tasmania), Russia (Urals), and Argentina. The most spectacular crystals sought by collectors come especially from Tasmania, where they form under particularly favorable supergene alteration conditions.
How do you recognize crocoite from other minerals?
Crocoite is easily recognized by its distinctive intense red-orange color, unique among common minerals and linked to its lead chromate composition (PbCrO₄). Its elongated prismatic crystals, vitreous luster, and high density (6.1) are additional distinctive features that make it unmistakable.
What is the value and price of crocoite in the collector's market?
The price of crocoite varies considerably based on the quality, size, and origin of the crystal, ranging from tens of euros for small samples to several thousand euros for museum-grade specimens. Transparent, well-formed crystals from Australian sources are the most sought-after and expensive on the mineralogical collector's market.
How is crocoite formed and under what geological conditions does it develop?
Crocoite forms through supergene oxidation processes (weathering) in the superficial environment of primary galena (PbS) deposits, when lead reacts with chromates derived from the alteration of chromium-bearing minerals. It represents an advanced stage of oxidation and is generally associated with secondary minerals such as cerussite (lead carbonate) and anglesite (lead sulfate).
Entry generated with Claude API (Anthropic) on data extracted from Mindat, RRUFF and Wikipedia. Not yet reviewed by a human expert. Verify data against original sources before citing in formal work.